Answer:
7ft (2.13 m).
Explanation:
From the question above, the following parameters/data are given; the room dimension = 12 ft × 18 ft (∼3.7 m × ∼5.5 m), the floor-to-ceiling height = 9 ft (2.8 m) and the spacing ratio for the luminaire is 1:0.
Note that, we are not given the value or data for the height of the plane, therefore, we will make an assumption that the height of the plane = 2ft.
Hence, the work plane to luminaire distance = (9 - 2)ft = 7ft (2.13 m).
So, the maximum distance that the luminaires can be separated and achieve uniform illuminance is;
= the work plane to luminaire distance × spacing ratio for the luminaire.
= 7ft (2.13 m) × 1 = 7ft (2.13 m).
Thus, the maximum distance that the luminaires can be separated and achieve uniform illuminance is 7ft (2.13 m).
Answer:
hello some parts of your question is missing attached below is the missing part
answer :
A) Determine changes in the 50-mm dimensions
The changes are : 0.006mm compression in y-direction
0.002 mm expansion in x and z directions
B) the stress required are evenly distributed
Explanation:
<em>Given data</em> :
50-mm cube of graphite fiber reinforced polymer matrix
subjected to 125-KN force in direction 2,
direction 2 is perpendicular to fiber direction ( direction 1 ) and cube is constrained against expansion in direction 3
A) Determine changes in the 50-mm dimensions
The changes are : 0.006mm compression in y-direction
0.002 mm expansion in x and z directions
B) the stress required are evenly distributed
attached below is the detailed solution
Answer:
C. Yes, and it is highly probable.
Explanation:
Null hypothesis, 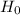 : ц 0.09
: ц 0.09
Alternative hypothesis, 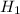 : ц > 0.09
: ц > 0.09
Test statistic is,
x Z = x - ц / s


=1
The p-value is,
p = P(Z > z)
=1—P(Z ≤1)
= 1— 0.841345
= 0.158655
(From normal tables)
The p-value is greater than the significance level, so we fail to reject the null hypothesis. The correct option is, C
Yes, and it is highly probable.
Answer:
I would say 10000;1 but thats just me
Explanation:
Answer:
Option A
hands-on assembly of the company's product
Explanation:
The managerial roles mainly include supervision of actual work on a higher level and implementation of organization's policy. The hands-on assembly of company's product is majorly done by junior staff who work under a supervisor who reports to the manager. Therefore, this is not a duty of a manager in a manufacturing field.