Answer:
Glycogen in an important storage polysaccharide found in animal tissues.
Explanation:
Full question:
Glycogen ________
A) forms the regulatory molecules known as enzymes
B) serves as a structural component of human cells
C) helps to protect vital organs from damage
D) is an important storage polysaccharide found in animal tissues
E) contains the genetic information found in cells
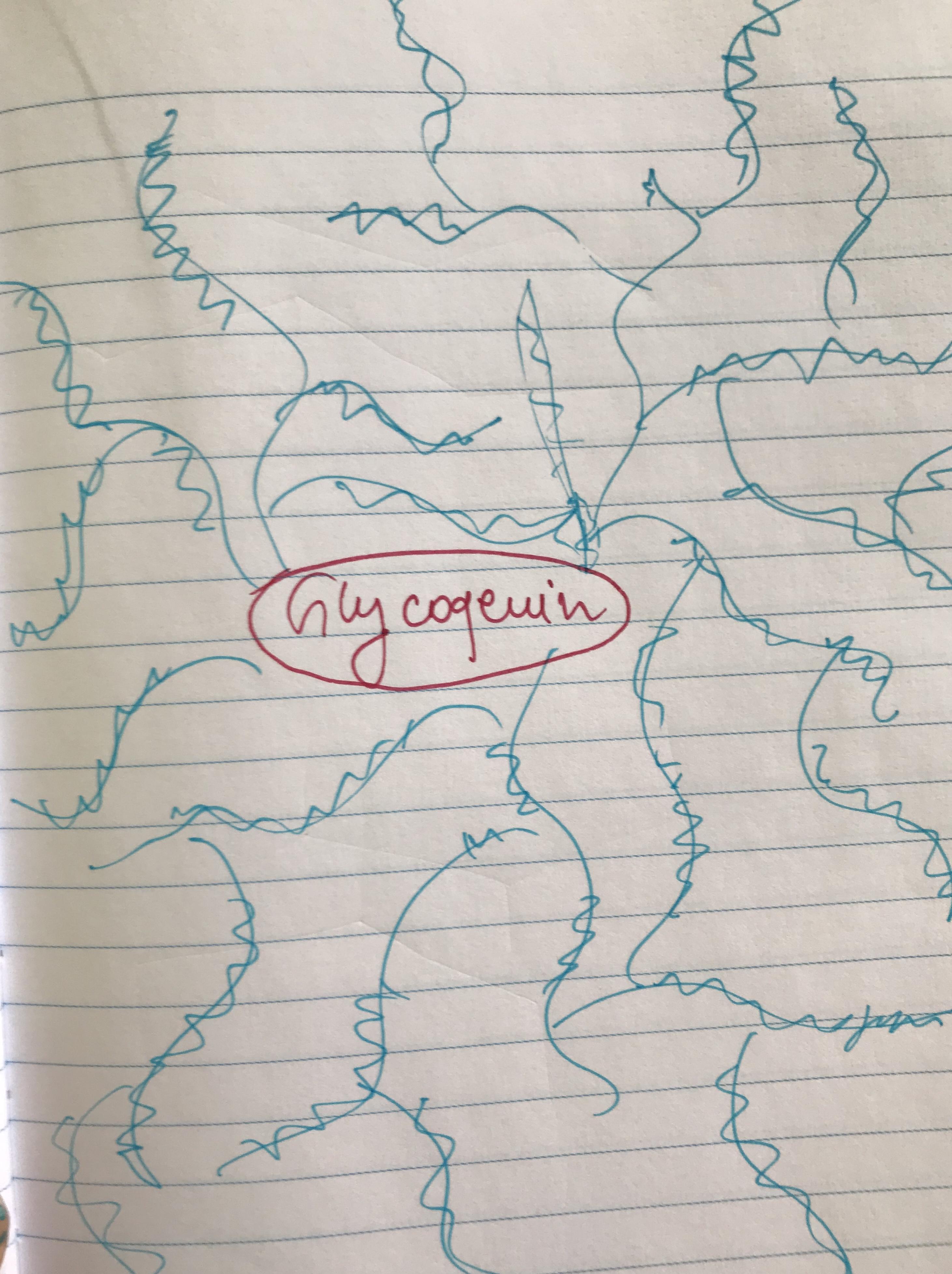
Glycogen is a complex polysaccharide of glucose founded in humans, animals, fungi and even bacteria. In humans, the glycogen is made and stored in liver cells. In the center on glycogen molecule, there is a single protein called Glycogenin. It is a center of a big flower made of glucose molecules (please refer to the scheme attached - Glycogenin is red and the blue lines are glucose chains). Glycogen is also stored in skeletal muscle, red and white blood cells, in glial brain cells and kidneys but in a smaller amounts. It can be found in the placenta in pregnant women where it serves as a nutrient storage for embryo. In an adult, the liver weighs 1,5 kg and glycogen weighs about 120g in such a liver. After a meal, the level of sugar is rising and the insulin is being secreted. Insulin is a tool by which sugar is being delivered to the cells, like a food delivery. During this period, glycogen is being synthesized in the liver out of glucose residues. When the meal is digested, the sugar level is back to normal. When more energy is needed, glycogen from the liver is broken down by glycogen phosphorylase and the new sugar is released into the bloodstream.
Answer:
d = 0.9 g/L
Explanation:
Given data:
Number of moles = 1 mol
Volume = 24.2 L
Temperature = 298 K
Pressure = 101.3 Kpa (101.3/101 = 1 atm)
Density of sample = ?
Solution:
PV = nRT (1)
n = number of moles
number of moles = mass/molar mass
n = m/M
Now we will put the n= m/M in equation 1.
PV = m/M RT (2)
d = m/v
PM = m/v RT ( by rearranging the equation 2)
PM = dRT
d = PM/RT
The molar mass of neon is = 20.1798 g/mol
d = 1 atm × 20.1798 g/mol / 0.0821 atm. L/mol.K × 273K
d = 20.1798 g/22.413 L
d = 0.9 g/L
Answer:
is cool is cool goodcripopo
Neon belongs to 2nd Period & 18th Group
The right answer for the question that is being asked and shown above is that: "C) carbon monoxide and carbon dioxide" hydrocarbons burn completely in an excess of oxygen, the products are <span>C) carbon monoxide and carbon dioxide</span>